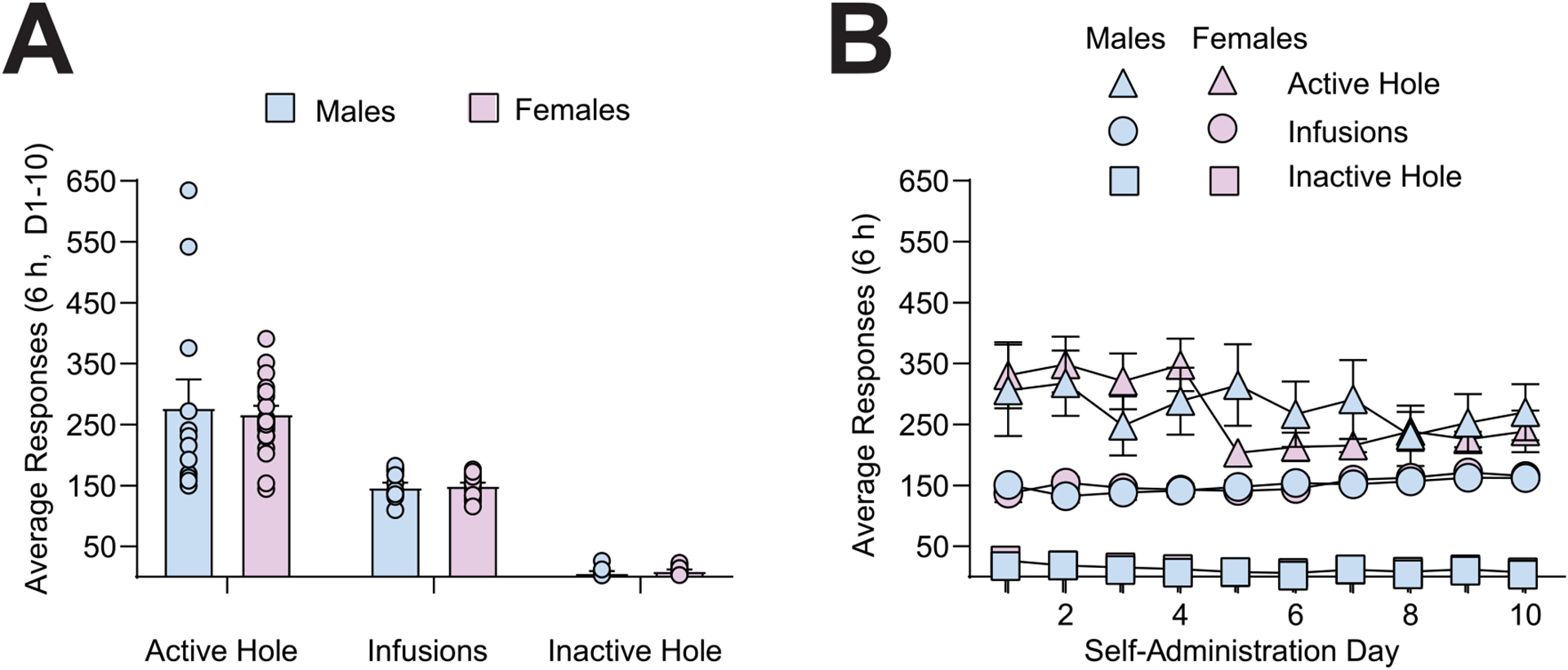
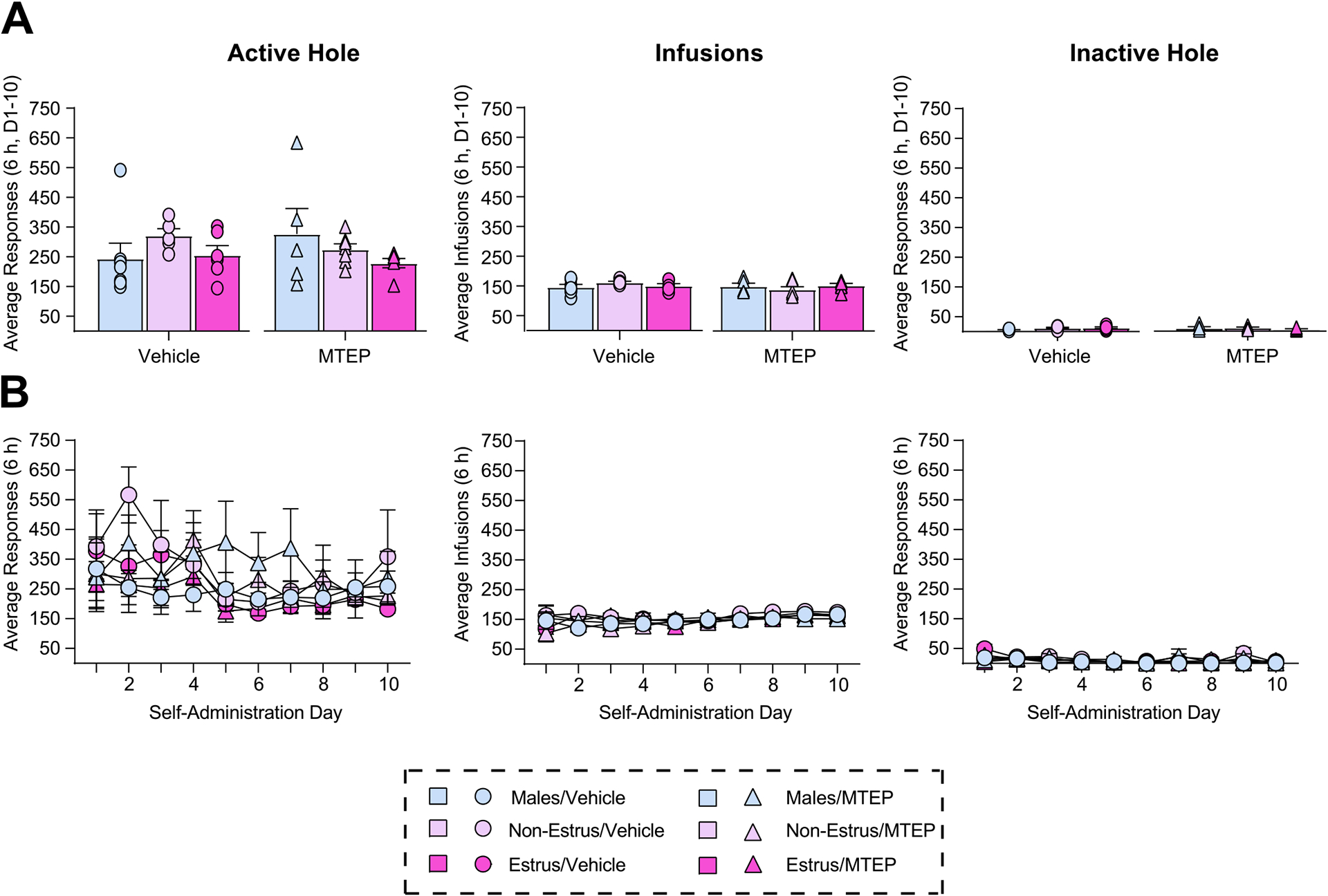
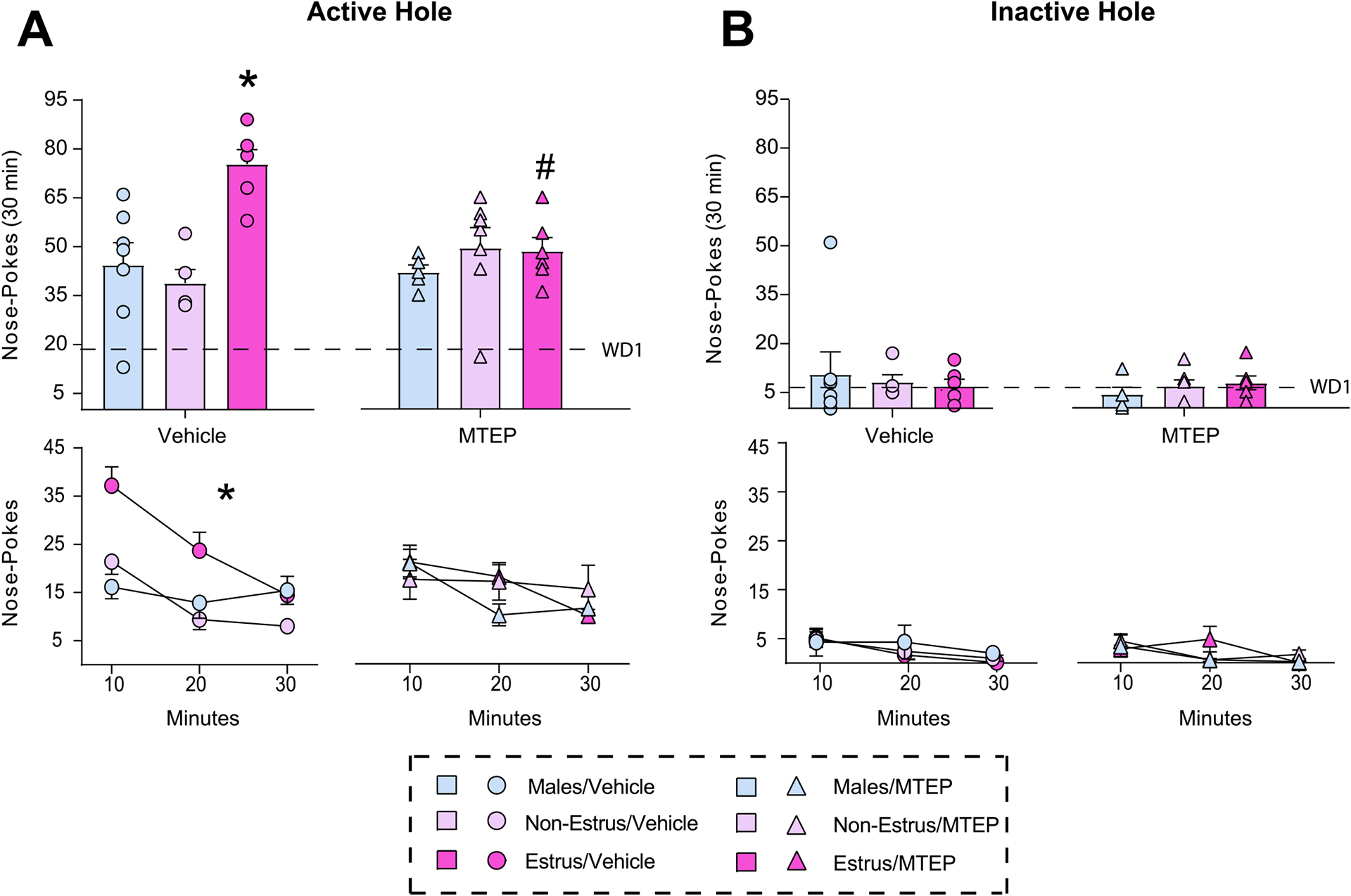
Drug associated cues are a common relapse trigger for individuals recovering from cocaine use disorder. Sex and ovarian hormones influence patterns of cocaine use and relapse vulnerability, with studies indicating that females show increased cue-induced craving and relapse vulnerability compared to males. In a rodent model of cocaine craving and relapse vulnerability, cue-induced cocaine seeking behavior following weeks of withdrawal from extended-access cocaine self-administration is higher in females in the estrus stage of the reproductive (estrous) cycle (Estrus Females) compared to both Males and females in all other stages (Non-Estrus Females). However, the neuronal substrates and cellular mechanisms underlying these sex differences is not fully understood. One region that contributes to both sex differences in behavioral responding and cue-induced cocaine seeking is the basolateral amygdala (BLA), while one receptor known to play a critical role in mediating cocaine seeking behavior is metabotropic glutamate receptor 5 (mGlu5). Here we assessed the effects of BLA mGlu5 inhibition following prolonged withdrawal from cocaine self-administration on observed estrous cycle-dependent changes in cue-induced cocaine seeking behavior. We found that BLA microinjections of the mGlu5 antagonist MTEP selectively reduced the enhanced cue-induced cocaine seeking normally observed in Estrus Females while having no effect on cocaine seeking in Males and Non-Estrus Females. These findings identify a unique interaction between cocaine-exposure, estrous cycle fluctuations and BLA mGlu5-dependent transmission on cue-induced cocaine seeking behavior.