The use of therapeutic apheresis (TA) either as stand-alone or adjunctive treatment in kidney transplantation has increased over the years to become a leading indication. This study shows recent trends in indications for TA related to kidney transplantation, adverse events, and patient outcome in this cohort.
This is a retrospective cohort review of adults who received TA for kidney transplant-related indications from January 1, 2017, to December 31, 2022, at the University of Virginia Medical Centre, Charlottesville, VA, USA. Data extracted include basic demographics, indication for apheresis, number of procedures, procedure characteristics, procedure-related adverse events (complications), and serum ionized calcium and serum creatinine. Data were analyzed using statistical package for social sciences (SPSS 2022 IBM Inc).
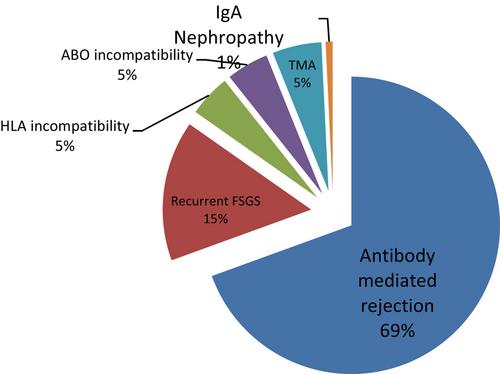
Data from a total of 131 patients who received 860 TA procedures were analyzed. Indications for TA were antibody-mediated rejection (65.5%), recurrent focal segmental glomerulosclerosis (15%), thrombotic microangiopathy (5%), desensitization for ABO incompatibility (4.5%) and for HLA-incompatibility (4.5%), and recurrent IgA nephropathy (1%). Some adverse events were encountered in 16.7% of the procedures and include hypocalcemia (7%), vascular access malfunction (0.7%), hypotension (1.2%), arrhythmia (0.6%), and depletion coagulopathy (0.6%). The overall case mortality rate was 8.4% over the 6-year period. There was one death recorded on machine during TA resulting in a procedure-mortality rate of 0.12%.
Antibody-mediated rejection was the most common indication for TA related to kidney transplantation. Adverse events were minor and patient survival over the time was within usual limits.