Previous proteomics studies in dysferlinopathy muscle have been limited in scope, often utilizing 2D-electrophoresis and yielding only a small number of differential expression calls. To address this gap, this study aimed to employ high-resolution proteomics to explore the proteomic landscapes of dysferlinopathy and analyze the correlation between muscle pathological changes and alterations in protein expression in muscle biopsies.
We conducted a comprehensive approach to investigate the proteomic profile and disease-associated changes in the muscle tissue proteome from 15 patients with dysferlinopathy, exhibiting varying degrees of dystrophic pathology, alongside age-matched controls. Our methodology encompasses tandem mass tag (TMT)-labeled liquid chromatography-mass spectrometry (LC–MS/MS)-based proteomics, protein–protein interaction (PPI) network analysis, weighted gene co-expression network analysis, and differential expression analysis. Subsequently, we examined the correlation between the expression of key proteins and the clinical characteristics of the patients to identify pathogenic targets associated with DYSF mutations in dysferlinopathy.
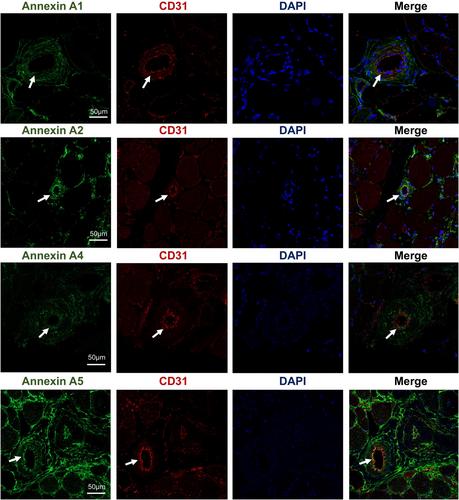
A total of 1600 differentially expressed proteins were identified, with 1321 showing high expression levels and 279 expressed at lower levels. Our investigation yields a molecular profile delineating the altered protein networks in dysferlinopathy-afflicted skeletal muscle, uncovering dysregulation across numerous cellular pathways and molecular processes, including mRNA metabolic processes, regulated exocytosis, immune response, muscle system processes, energy metabolic processes, and calcium transmembrane transport. Moreover, we observe significant associations between the protein expression of ANXA1, ANXA2, ANXA4, ANXA5, LMNA, PYGM, and the extent of histopathologic changes in muscle biopsies from patients with dysferlinopathy, validated through immunoblotting and immunofluorescence assays.
Through the aggregation of expression data from dysferlinopathy-impacted muscles exhibiting a range of pathological alterations, we identified multiple key proteins associated with the dystrophic pathology of patients with dysferlinopathy. These findings provide novel insights into the pathogenesis of dysferlinopathy and propose promising targets for future therapeutic endeavors.