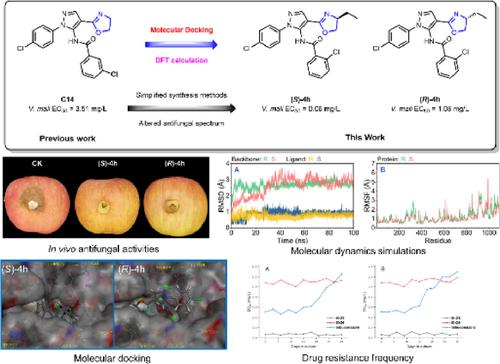
Development of novel chiral antifungal agents for effective control of plant pathogens is urgently needed. In this study, a series of pyrazol-5-yl-benzamide derivatives containing chiral oxazoline moiety were rationally designed and developed based on molecular docking.
The in vitro antifungal assay results indicated that compounds (rac)-4h (R1 = Et), (S)-4 h (R1 = S-Et) and (R)-4 h (R1 = R-Et) exhibited remarkable antifungal activities against Valsa mali with median effective concentration (EC50) values of 0.24, 0.06 and 1.08 mg/L, respectively. Preliminary structure–activity relationships (SARs) revealed that the modification of the chiral substituent group at the oxazoline moiety significantly affected the antifungal activities of the target compounds. Furthermore, compounds (S)-4h (87.5%) and (R)-4h (84.3%) exhibited in vivo protective activities comparable to tebuconazole (87.5%) against V. mali. Subsequent molecular docking analysis, succinate dehydrogenase (SDH) enzyme inhibition assays and molecular dynamic (MD) simulations verified that the potential target enzyme of this class of derivatives could be SDH and helped to explain the large difference in antifungal activities of compounds (S)-4h and (R)-4h. Confocal laser scanning microscopy (CLSM) and scanning electron microscopy (SEM) observations confirmed that these two compounds severely disrupted the mycelial morphology of V. mali. Theoretical calculation studies provided some insight into the subsequent modification of such pyrazol-5-yl-benzamide derivatives. Resistance frequency studies showed that (S)-4h and (R)-4h treatments were less likely to produce resistant fungal strains than tebuconazole. Meanwhile, compounds (S)-4h and (R)-4h exhibited no apparent toxicity to the Apis mellifera L. population.
Therefore, these derivatives are potential candidates for the development of novel chiral fungicides for crop protection. © 2025 Society of Chemical Industry.