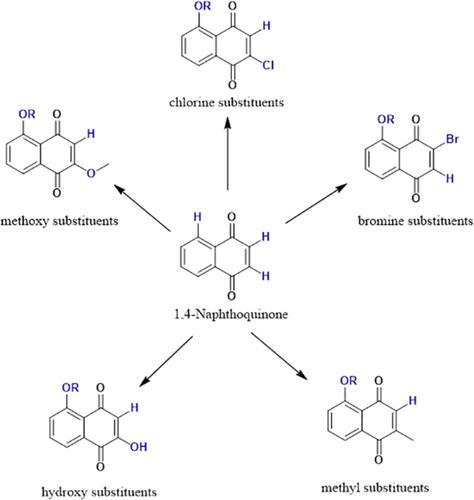
1,4-Naphthoquinone compounds have shown pesticidal activity against Aedes aegypti larvae, a key vector of diseases such as dengue and Zika. However, limited knowledge of their structure–activity relationships has hindered their optimization for pesticide development. This study investigates the structure–activity relationships of 1,4-naphthoquinone, particularly 2-hydroxy-1,4-naphthoquinone and its derivatives.
A series of 1,4-naphthoquinones with modifications at the C-2, C-5, and C-8 positions were synthesized and tested for larvicidal activities against Ae. aegypti. The presence of chlorine substituents at the C-2 position significantly increased mosquito larval mortality. In contrast, 2-hydroxy and 2-methoxy groups were less effective, highlighting the importance of the C-2 substituent in determining larvicidal activity. The compounds were tested on both pyrethroid-susceptible Orlando 1952 (ORL1952) and permethrin-resistant Puerto Rico (PR) strains of Ae. aegypti. While compound 1d exhibited the lowest median lethal concentration (LC50) against ORL1952 larvae, it showed minimal activity against the PR strain, which is resistant. The most promising compounds, 1 and 4a, showed LC50 values of 2.764 and 4.916 ppm in the ORL strain, and 2.548 and 4.878 ppm in the PR strain, respectively.
Chlorine substitution at the C-2 position of 1,4-naphthoquinones enhances larvicidal activity against Ae. aegypti, while 2-hydroxy and 2-methoxy substitutions reduce efficacy. Certain compounds, such as 1 and 4a, show potential as broad-spectrum agents effective against both susceptible and resistant strains of Ae. aegypti, highlighting their promise for further development as mosquito control agents. © 2025 Society of Chemical Industry.