Plant pathogenic fungi are a major contributor to reductions in crop yield and quality, posing significant challenges to global food security. The extensive application of chemical fungicides has led to the development of resistance in pathogenic fungi and the accumulation of harmful residues, which threaten environmental sustainability and human health. Plant-derived fungicides, with low toxicity and broad-spectrum activity, offer an eco-friendly alternative to synthetic chemicals.
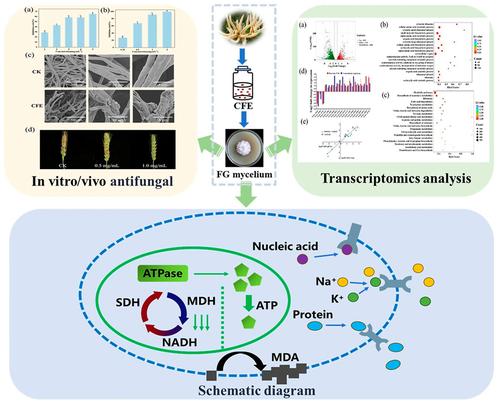
This study evaluated the antifungal activity of chestnut flower extract (CFE) against Fusarium graminearum (FG) and investigated its underlying mechanisms through cellular and transcriptomic analyses. Liquid chromatography-mass spectrometry (LC–MS) identified phenolic acids and flavonoids as the primary active constituents of CFE. CFE inhibited mycelial growth and spore germination with median effective concentration (EC50) values of 2.51 and 0.39 mg mL−1, respectively. It disrupted biofilm integrity and membrane permeability by reducing ergosterol content, increasing extracellular conductivity, and affecting malondialdehyde (MDA) levels. Protein and nucleic acid leakage were observed. Additionally, CFE inhibited energy metabolism by reducing adenosine triphosphate (ATP) levels and suppressing the activities of key respiratory enzymes, including succinate dehydrogenase (SDH), malate dehydrogenase (MDH) and nicotinamide adenine dinucleotide (NADH) dehydrogenase. Transcriptomic analysis further revealed that CFE affected multiple biological processes in FG, including cell structure, protein synthesis, ion transport and mitochondrial function.
The chestnut flower contains active antibacterial ingredients that exhibit targeted inhibition of FG, thereby providing theoretical and technical support for the development of natural antibacterial agents with specific targeting capabilities. © 2025 Society of Chemical Industry.