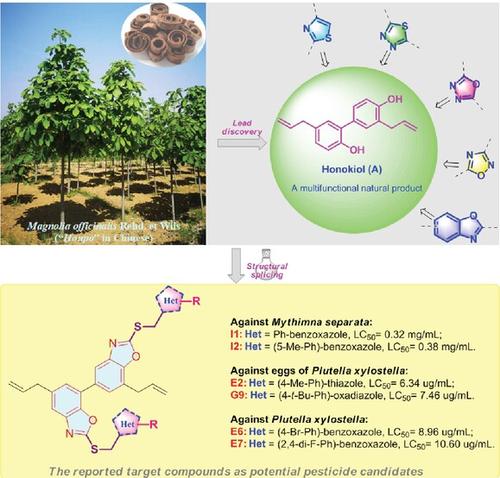
Persistent and excessive application of conventional synthetic pesticides has triggered a series of environmental and agricultural product safety risks in recent decades. Natural products are considered to provide considerable sources for the innovation of novel ecofriendly agrochemicals with high efficacy.
In this work, a structural splicing strategy was used to prepare 37 honokiol-derived thioether analogs bearing different heterocyclic scaffolds and their pesticidal activities were assessed. Bioassay results showed that most of the target compounds have growth inhibitory activity against Mythimna separata. Notably, compounds I1 and I2 showed prominent growth inhibitory activity against M. separata with 50% lethal concentration (LC50) values of 0.32 and 0.38 mg mL−1, respectively, which were superior to precursor honokiol (LC50 = 1.46 mg mL−1) and commercial insecticide toosendanin (LC50 = 0.63 mg mL−1). Compounds E2 and G9 demonstrated pronounced ovicidal activities against the eggs of Plutella xylostella with LC50 values of 6.34 and 7.49 μg mL−1, respectively, compared with the LC50 value of honokiol of 46.96 μg mL−1. Furthermore, seven compounds E4, E6, E7, E8, G3, H5, and I5 showed excellent larvicidal activities against P. xylostella, significantly outperforming honokiol and the positive control rotenone. Preliminary analysis of structure–activity relationships reveals that the heterocyclic moieties introduced are indeed beneficial for the insecticidal potential of these compounds.
The novel scaffolds and remarkable insecticidal potency position the honokiol-based analogs reported herein as promising candidate leads for further development as agrochemicals for pest management. © 2025 Society of Chemical Industry.