Muscle wasting is a debilitating co-morbidity affecting most advanced cancer patients. Alongside enhanced muscle catabolism, defects in muscle repair/regeneration contribute to cancer-associated wasting. Among the factors implicated in suppression of muscle regeneration are cytokines that interfere with myogenic signal transduction pathways. Less understood is how other cancer/wasting-associated cues, such as metabolites, contribute to muscle dysfunction. This study investigates how the metabolite succinate affects myogenesis and muscle regeneration.
We leveraged an established ectopic metabolite treatment (cell permeable dimethyl-succinate) strategy to evaluate the ability of intracellular succinate elevation to (i) affect myoblast homeostasis (proliferation and apoptosis), (ii) disrupt protein dynamics and induce wasting-associated atrophy, and (iii) modulate in vitro myogenesis. In vivo succinate supplementation experiments (2% succinate and 1% sucrose vehicle) were used to corroborate and extend in vitro observations. Metabolic profiling and functional metabolic studies were then performed to investigate the impact of succinate elevation on mitochondria function.
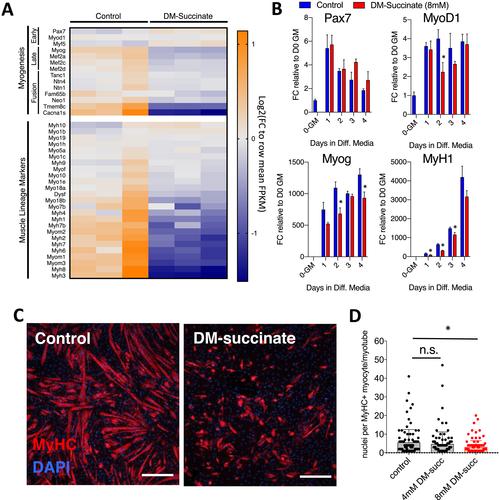
We found that in vitro succinate supplementation elevated intracellular succinate about 2-fold and did not have an impact on proliferation or apoptosis of C2C12 myoblasts. Elevated succinate had minor effects on protein homeostasis (~25% decrease in protein synthesis assessed by O-propargyl-puromycin staining), and no significant effect on myotube atrophy. Succinate elevation interfered with in vitro myoblast differentiation, characterized by significant decreases in late markers of myogenesis and fewer nuclei per myosin heavy chain positive structure (assessed by immunofluorescence staining). While mice orally administered succinate did not exhibit changes in overall body composition or whole muscle weights, these mice displayed smaller muscle myofiber diameters (~6% decrease in the mean of non-linear regression curves fit to the histograms of minimum feret diameter distribution), which was exacerbated when muscle regeneration was induced with barium chloride injury. Significant decreases in the mean of non-linear regression curves fit to the histograms of minimum feret diameter distributions were observed 7 and 28 days post injury. Elevated numbers of myogenin positive cells (three-fold increase) supportive of the differentiation defects observed in vitro were observed 28 days post injury. Metabolic profiling and functional metabolic assessment of myoblasts revealed that succinate elevation caused both widespread metabolic changes and significantly lowered maximal cellular respiration (~35% decrease).
This study broadens the repertoire of wasting-associated factors that can directly modulate muscle progenitor cell function and strengthens the hypothesis that metabolic derangements are significant contributors to impaired muscle regeneration, an important aspect of cancer-associated muscle wasting.