下载PDF
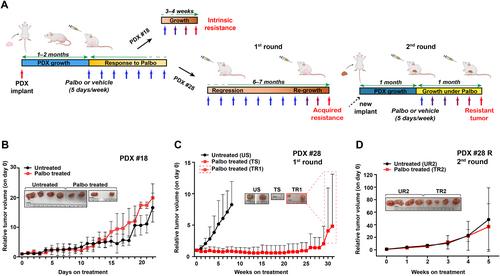
{"title":"用于捕捉肿瘤异质性和探索 CDK4/6 抑制剂抗药性机制的综合腔隙性乳腺癌患者衍生异种移植物 (PDX) 库。","authors":"Ilenia Segatto, Maria Chiara Mattevi, Gian Luca Rampioni Vinciguerra, Nicole Crestan, Lorena Musco, Andrea Favero, Alessandra Dall'Acqua, Gabriele Di Giustino, Giorgia Mungo, Sara D'Andrea, Chiara Gava, Federica Ruggiero, Matteo Dugo, Lorenzo Gerratana, Fabio Puglisi, Samuele Massarut, Riccardo Bomben, Maurizio Callari, Tiziana Perin, Gustavo Baldassarre, Barbara Belletti","doi":"10.1002/path.6358","DOIUrl":null,"url":null,"abstract":"<p>Breast cancer (BC) is marked by significant genetic, morphological and clinical heterogeneity. To capture this heterogeneity and unravel the molecular mechanisms driving tumor progression and drug resistance, we established a comprehensive patient-derived xenograft (PDX) biobank, focusing particularly on luminal (estrogen receptor, ER+) and young premenopausal patients, for whom PDX models are currently scarce. Across all BC subtypes, our efforts resulted in an overall success rate of 17% (26 established PDX lines out of 151 total attempts), specifically 15% in luminal, 12% in human epidermal growth factor receptor 2 positive (HER2+) and 35% in triple negative BC. These PDX mirrored morphologic and genetic features of BC from which they originated, serving as a reliable tool to investigate drug resistance and test therapeutic strategies. We focused on understanding resistance to CDK4/6 inhibitors (CDK4/6i), which are crucial in the treatment of patients with advanced luminal BC. Treating a sensitive luminal BC PDX with the CDK4/6i palbociclib revealed that, despite initial tumor shrinkage, some tumors might eventually regrow under drug treatment. RNA sequencing, followed by gene set enrichment analyses, unveiled that these PDXs have become refractory to CDK4/6i, both at biological and molecular levels, displaying significant enrichment in proliferation pathways, such as <i>MTORC1</i>, <i>E2F</i> and <i>MYC</i>. Using organoids derived from these PDX (PDxO), we observed that acquisition of CDK4/6i resistance conferred cross-resistance to endocrine therapy and that targeting MTORC1 was a successful strategy to overcome CDK4/6i resistance. Considered together, these results indicate that our PDX models may serve as robust tools to elucidate the molecular basis of BC disease progression and, by providing the possibility to simultaneously test different therapies on the same tumor, to surmount treatment resistance. While this approach is of course not feasible in the clinic, its exploitation in PDX may expedite the identification and development of more successful therapies for patients with advanced luminal BC. © 2024 The Author(s). <i>The Journal of Pathology</i> published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.</p>","PeriodicalId":232,"journal":{"name":"The Journal of Pathology","volume":"264 4","pages":"434-447"},"PeriodicalIF":5.6000,"publicationDate":"2024-10-25","publicationTypes":"Journal Article","fieldsOfStudy":null,"isOpenAccess":false,"openAccessPdf":"https://onlinelibrary.wiley.com/doi/epdf/10.1002/path.6358","citationCount":"0","resultStr":"{\"title\":\"A comprehensive luminal breast cancer patient-derived xenografts (PDX) library to capture tumor heterogeneity and explore the mechanisms of resistance to CDK4/6 inhibitors\",\"authors\":\"Ilenia Segatto, Maria Chiara Mattevi, Gian Luca Rampioni Vinciguerra, Nicole Crestan, Lorena Musco, Andrea Favero, Alessandra Dall'Acqua, Gabriele Di Giustino, Giorgia Mungo, Sara D'Andrea, Chiara Gava, Federica Ruggiero, Matteo Dugo, Lorenzo Gerratana, Fabio Puglisi, Samuele Massarut, Riccardo Bomben, Maurizio Callari, Tiziana Perin, Gustavo Baldassarre, Barbara Belletti\",\"doi\":\"10.1002/path.6358\",\"DOIUrl\":null,\"url\":null,\"abstract\":\"<p>Breast cancer (BC) is marked by significant genetic, morphological and clinical heterogeneity. To capture this heterogeneity and unravel the molecular mechanisms driving tumor progression and drug resistance, we established a comprehensive patient-derived xenograft (PDX) biobank, focusing particularly on luminal (estrogen receptor, ER+) and young premenopausal patients, for whom PDX models are currently scarce. Across all BC subtypes, our efforts resulted in an overall success rate of 17% (26 established PDX lines out of 151 total attempts), specifically 15% in luminal, 12% in human epidermal growth factor receptor 2 positive (HER2+) and 35% in triple negative BC. These PDX mirrored morphologic and genetic features of BC from which they originated, serving as a reliable tool to investigate drug resistance and test therapeutic strategies. We focused on understanding resistance to CDK4/6 inhibitors (CDK4/6i), which are crucial in the treatment of patients with advanced luminal BC. Treating a sensitive luminal BC PDX with the CDK4/6i palbociclib revealed that, despite initial tumor shrinkage, some tumors might eventually regrow under drug treatment. RNA sequencing, followed by gene set enrichment analyses, unveiled that these PDXs have become refractory to CDK4/6i, both at biological and molecular levels, displaying significant enrichment in proliferation pathways, such as <i>MTORC1</i>, <i>E2F</i> and <i>MYC</i>. Using organoids derived from these PDX (PDxO), we observed that acquisition of CDK4/6i resistance conferred cross-resistance to endocrine therapy and that targeting MTORC1 was a successful strategy to overcome CDK4/6i resistance. Considered together, these results indicate that our PDX models may serve as robust tools to elucidate the molecular basis of BC disease progression and, by providing the possibility to simultaneously test different therapies on the same tumor, to surmount treatment resistance. While this approach is of course not feasible in the clinic, its exploitation in PDX may expedite the identification and development of more successful therapies for patients with advanced luminal BC. © 2024 The Author(s). <i>The Journal of Pathology</i> published by John Wiley & Sons Ltd on behalf of The Pathological Society of Great Britain and Ireland.</p>\",\"PeriodicalId\":232,\"journal\":{\"name\":\"The Journal of Pathology\",\"volume\":\"264 4\",\"pages\":\"434-447\"},\"PeriodicalIF\":5.6000,\"publicationDate\":\"2024-10-25\",\"publicationTypes\":\"Journal Article\",\"fieldsOfStudy\":null,\"isOpenAccess\":false,\"openAccessPdf\":\"https://onlinelibrary.wiley.com/doi/epdf/10.1002/path.6358\",\"citationCount\":\"0\",\"resultStr\":null,\"platform\":\"Semanticscholar\",\"paperid\":null,\"PeriodicalName\":\"The Journal of Pathology\",\"FirstCategoryId\":\"3\",\"ListUrlMain\":\"https://onlinelibrary.wiley.com/doi/10.1002/path.6358\",\"RegionNum\":2,\"RegionCategory\":\"医学\",\"ArticlePicture\":[],\"TitleCN\":null,\"AbstractTextCN\":null,\"PMCID\":null,\"EPubDate\":\"\",\"PubModel\":\"\",\"JCR\":\"Q1\",\"JCRName\":\"ONCOLOGY\",\"Score\":null,\"Total\":0}","platform":"Semanticscholar","paperid":null,"PeriodicalName":"The Journal of Pathology","FirstCategoryId":"3","ListUrlMain":"https://onlinelibrary.wiley.com/doi/10.1002/path.6358","RegionNum":2,"RegionCategory":"医学","ArticlePicture":[],"TitleCN":null,"AbstractTextCN":null,"PMCID":null,"EPubDate":"","PubModel":"","JCR":"Q1","JCRName":"ONCOLOGY","Score":null,"Total":0}
引用次数: 0
引用
批量引用