The earliest manifestation of alcohol-associated liver disease (ALD) is steatosis characterized by deposition of fat in specialized organelles called lipid droplets (LDs). While alcohol administration causes a rise in LD numbers in the hepatocytes, little is known regarding their characteristics that allow their accumulation and size to increase. The aim of the present study is to gain insights into underlying pathophysiological mechanisms by investigating the ethanol-induced changes in hepatic LD proteome as a function of LD size.
Adult male Wistar rats (180–200 g BW) were fed with ethanol liquid diet for 6 weeks. At sacrifice, large-, medium-, and small-sized hepatic LD subpopulations (LD1, LD2, and LD3, respectively) were isolated and subjected to morphological and proteomic analyses.
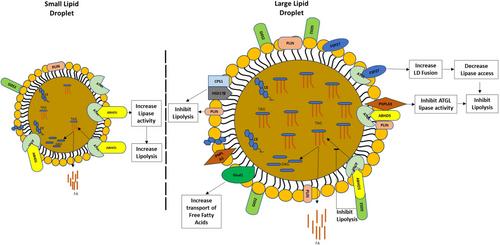
Morphological analysis of LD1-LD3 fractions of ethanol-fed rats clearly demonstrated that LD1 contained larger LDs compared with LD2 and LD3 fractions. Our preliminary results from principal component analysis showed that the proteome of different-sized hepatic LD fractions was distinctly different. Proteomic data analysis identified over 2000 proteins in each LD fraction with significant alterations in protein abundance among the three LD fractions. Among the altered proteins, several were related to fat metabolism, including synthesis, incorporation of fatty acid, and lipolysis. Ingenuity pathway analysis revealed increased fatty acid synthesis, fatty acid incorporation, LD fusion, and reduced lipolysis in LD1 compared to LD3. Overall, the proteomic findings indicate that the increased level of protein that facilitates fusion of LDs combined with an increased association of negative regulators of lipolysis dictates the generation of large-sized LDs during the development of alcohol-associated hepatic steatosis.
Several significantly altered proteins were identified in different-sized LDs isolated from livers of ethanol-fed rats. Ethanol-induced increases in specific proteins that hinder LD lipid metabolism led to the accumulation and persistence of large-sized LDs in the liver.