Sinonasal undifferentiated carcinoma (SNUC) is a rare and highly aggressive malignancy originating in the nasal cavity and paranasal sinuses. Its pathogenesis and immune characteristics remain poorly understood.
This study investigates the molecular aspects of SNUC, focusing on tumorigenesis and immunity.
For this purpose, spatial transcriptome analysis was employed to compare the gene expression profiles of SNUC tumor cells with those of normal epithelial cells, as well as to compare tumor-infiltrating immune cells with immune cells from normal, tumor-free tissue areas. For validation, next-generation sequencing tests and clinical sample studies were conducted.
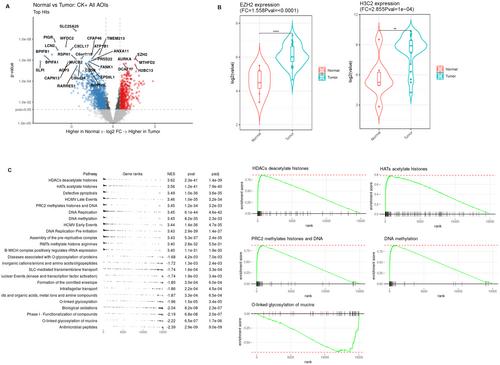
Spatial transcriptome analysis revealed notable upregulation of EZH2 and the histone family gene such as H3C2 (H3-clustered histone 2) in SNUC tumor cells. Additionally, gene set enrichment analysis identified significant activations in the histone deacetylase (HDAC) signaling pathway, histone acetyltransferase (HAT) pathway, polycomb repressive complex 2 (PRC2), and DNA methylation pathways. A notable decrease was observed in downregulated genes and pathways, including the mucin family of protein genes, the keratin protein gene, and the mucin glycosylation pathway. Next-generation sequencing did not reveal specific genetic mutations within these pathways, although mutations such as IDH2 R172S were noted. Clinical SNUC tissues confirmed increased immunoexpression of EZH2 and PRC2 markers. Analysis of tumor immunity revealed a characteristic immune cell signature, with a notable predominance of naïve B cells, macrophages, CD8 memory T cells, and Tregs in the SNUC microenvironment, alongside the increased expression of LAG3 in tumor-infiltrating immune cells.
Our study suggests epigenetic mechanisms, particularly via EZH2, play a crucial role in SNUC carcinogenesis. Furthermore, distinctive immune cell profiles in SNUC point to potential immune-related characteristics of this malignancy.