Obesity has been linked to a more severe phenotype in patients with ulcerative colitis (UC).
To evaluate the impact of obesity on outcomes of advanced therapies in UC.
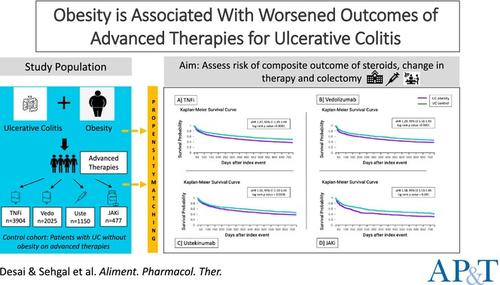
We conducted a retrospective cohort study utilising the TriNetX database comparing the composite score of corticosteroid use, change in advanced therapy or colectomy within two years between two cohorts of patients with UC—those with obesity (BMI ≥ 30 kg/m2) and those without (BMI 18.5–24.9). The risk assessment was stratified to specific advanced therapies, including tumour necrosis factor α inhibitors (TNFi), vedolizumab, ustekinumab and Janus kinase inhibitors (JAKi). We performed 1:1 propensity score matching (PSM) for demographics, co-morbid conditions, laboratory values and IBD medications including corticosteroids.
There were 3904, 2025, 1150 and 477 patients on TNFi, vedolizumab, ustekinumab and JAKi, respectively, in the UC obesity cohort. After PSM, the UC obesity cohort was at an increased risk of the composite outcome of corticosteroid use, change in therapy and colectomy compared to the UC control cohort in patients on TNFi (aHR 1.37, 95% CI 1.29–1.49), vedolizumab (aHR 1.29, 95% CI 1.16–1.43), ustekinumab (aHR 1.1.26, 95% CI 1.10–1.44) and JAKi (aHR 1.38, 95% CI 1.13–1.69). Sub-group analysis based on the specific TNFi also showed an increased risk of composite outcome for infliximab (aHR 1.36, 95% CI 1.22–1.52) and adalimumab (aHR 1.26, 95% CI 1.11–1.42) within 2 years.
Obesity is associated with lower efficacy of several advanced therapies in patients with UC.