While it is widely accepted that intravenous (IV) iron improves functional capacity, symptoms, and cardiovascular outcomes in patients with heart failure (HF) with reduced ejection fraction (HFrEF) diagnosed with iron deficiency (ID), three recently published cardiovascular outcome trials (AFFIRM-AHF, IRONMAN and HEART-FID) of IV iron supplementation in HF failed to demonstrate a significant benefit on their respective primary endpoints. Dosing of IV iron after the initial correction of baseline ID – by design or as a result of trial circumstances – was relatively low (i.e. <500 mg/year). The primary objective of the FAIR-HF2 trial is to evaluate the treatment effect of ferric carboxymaltose (FCM) compared with placebo in ambulatory patients with HFrEF using a higher dose of IV iron during follow-up (i.e. >1000 mg/year). The second objective of the study is to create prospective evidence for patients fulfilling the new definition of ID for patients with HF, i.e. for those with a transferrin saturation <20%.
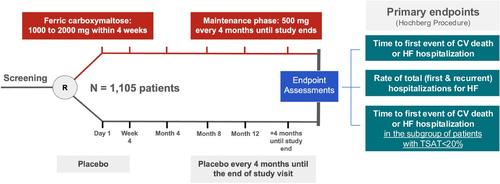
FAIR-HF2 is an investigator-initiated, multicentre, randomized, double-blind, placebo-controlled trial that has recruited 1105 patients with chronic HF with a left ventricular ejection fraction of ≤45% and concomitant ID, defined as serum ferritin <100 ng/ml or serum ferritin 100–299 ng/ml with a transferrin saturation <20%. Patients were consented and randomized to receive either IV FCM (treatment) or saline (placebo). During an estimated median follow-up of over 2 years, patients underwent a placebo-controlled repletion and maintenance phase, with an initial iron supplementation of up to 2000 mg, followed by 500 mg every 4 months unless stop criteria of haemoglobin >16 mg/dl or serum ferritin >800 ng/ml are met on repeat visits. The trial will evaluate three primary hypotheses: (i) time to first event of cardiovascular death or hospitalization for HF, (ii) the rate of total (first and recurrent) HF hospitalizations (both analysed in the full study population), and (iii) the time to first event of cardiovascular death or hospitalization for HF in patients with a transferrin saturation <20% at baseline. The familywise type I error rate across the three primary endpoint hypotheses will be controlled using the Hochberg procedure (alpha 0.05).
The FAIR-HF2 will evaluate the efficacy of FCM in patients with HFrEF in improving cardiovascular outcomes by utilizing a more aggressive approach towards iron supplementation ensuring prevention of transitional ID after initial repletion targets have been met.